Tautomeric influence on the photoinduced birefringence of 4-substituted phthalimide 2-hydroxy Schiff bases in PMMA matrix | SpringerLink

Tautomers and Rotamers of Curcumin: A Combined UV Spectroscopy, High-Performance Liquid Chromatography, Ion Mobility Mass Spectrometry, and Electronic Structure Theory Study | The Journal of Physical Chemistry A

Photoswitchable Keto–Enol Tautomerism Driven by Light-Induced Change in Antiaromaticity | Organic Letters

Photoinduced and ground state conversions in a cyclic β-thioxoketone - RSC Advances (RSC Publishing) DOI:10.1039/D1RA09020H
![Energies | Free Full-Text | Using Chou's 5-Step Rule to Evaluate the Stability of Tautomers: Susceptibility of 2-[(Phenylimino)-methyl]-cyclohexane-1,3-diones to Tautomerization Based on the Calculated Gibbs Free Energies Energies | Free Full-Text | Using Chou's 5-Step Rule to Evaluate the Stability of Tautomers: Susceptibility of 2-[(Phenylimino)-methyl]-cyclohexane-1,3-diones to Tautomerization Based on the Calculated Gibbs Free Energies](https://pub.mdpi-res.com/energies/energies-13-00183/article_deploy/html/images/energies-13-00183-ag-550.jpg?1579175166)
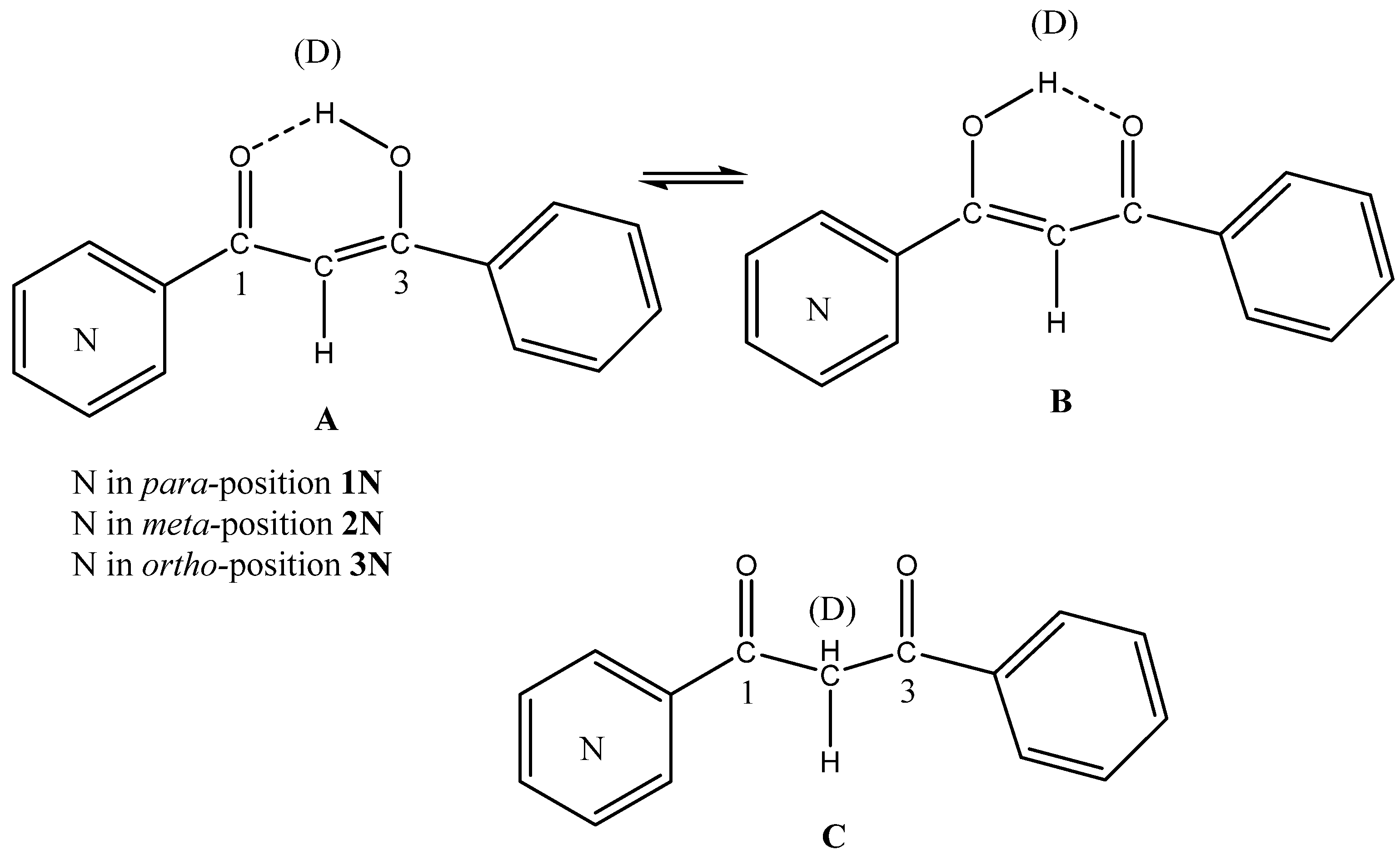
Energies | Free Full-Text | Using Chou's 5-Step Rule to Evaluate the Stability of Tautomers: Susceptibility of 2-[(Phenylimino)-methyl]-cyclohexane-1,3-diones to Tautomerization Based on the Calculated Gibbs Free Energies

Coupling carbon nanomaterials with photochromic molecules for the generation of optically responsive materials | Nature Communications

Synthesis and Photochromism of Novel Pyridyl-Substituted Naphthopyrans | The Journal of Organic Chemistry

Reversible Shifting of a Chemical Equilibrium by Light: The Case of Keto–Enol Tautomerism of a β-Ketoester | Organic Letters

Keto-enol tautomerization and intermolecular proton transfer in photoionized cyclopentanone dimer in the gas phase: The Journal of Chemical Physics: Vol 141, No 4

Controlling Keto–Enol Tautomerism of Ureidopyrimidinone to Generate a Single-Quadruple AADD-DDAA Dimeric Array | Organic Letters